Are you preparing for the RRB Group D exam? It is a huge opportunity to get a secure government job in Indian Railways. But to get selected, you need to score high marks in the Computer-Based Test (CBT). One section that troubles almost every student is General Science. And within Science, Chemistry is often the scariest part. Students get confused by complex formulas, atomic numbers, and chemical reactions, but they don’t know that they don’t need to be a scientist to clear this exam. RRB asks very basic Chemistry questions for RRB Group D exam that are mainly from Class 9 and 10 NCERT books. They focus on things we see in daily life—like why lemons are sour or what is inside a battery. To help you prepare without fear, we have compiled the most repeated Chemistry questions into a free PDF. You can download it from this blog.
Why is Chemistry Important for RRB Group D?
In the General Science section (25 questions), Chemistry plays a massive role.
- High Weightage: You can expect 8 to 10 questions purely from Chemistry.
- Direct & Factual: Unlike Physics, where you have to solve numerical problems, Chemistry questions are direct. Example: “What is the chemical name of Salt?”
- Rank Decider: Most students ignore Chemistry. If you prepare it well, you will automatically get ahead of lakhs of competitors.
Download Free PDF of Chemistry Questions for RRB Group D
We have prepared a comprehensive PDF containing exam-level questions. We have chosen questions that cover the unique mix of Periodic Table, Acids & Bases, and Metals that the RRB Group D is famous for. Download it, save it, and practice these questions to build your confidence.
Q1. Which gas is predominantly responsible for the “Greenhouse Effect” on Earth?
(a) Oxygen
(b) Nitrogen
(c) Carbon Dioxide
(d) Argon
Answer: (c) Carbon Dioxide
Logic:
Carbon dioxide (CO₂) traps heat in the Earth’s atmosphere, leading to an increase in global temperature, known as global warming. Methane is another important greenhouse gas, but CO₂ is the primary contributor.
Q2. What is the chemical formula of ‘Baking Soda’?
(a) Na₂CO₃
(b) NaHCO₃
(c) NaCl
(d) NaOH
Answer: (b) NaHCO₃
Logic:
Sodium Bicarbonate (NaHCO₃) is known as Baking Soda. Sodium Carbonate (Na₂CO₃) is Washing Soda. Students often confuse these two, so this distinction is important.
Q3. In the Modern Periodic Table, elements are arranged according to their:
(a) Atomic Mass
(b) Atomic Number
(c) Density
(d) Melting Point
Answer: (b) Atomic Number
Logic:
Mendeleev originally arranged elements according to atomic mass, but Henry Moseley later corrected this by arranging elements based on atomic number, which is followed in the Modern Periodic Table.
Q4. Which metal is liquid at room temperature?
(a) Sodium
(b) Iron
(c) Mercury
(d) Aluminum
Answer: (c) Mercury
Logic:
Mercury (Hg) is the only metal that remains liquid at room temperature. Bromine is the only non-metal that is liquid under the same conditions.
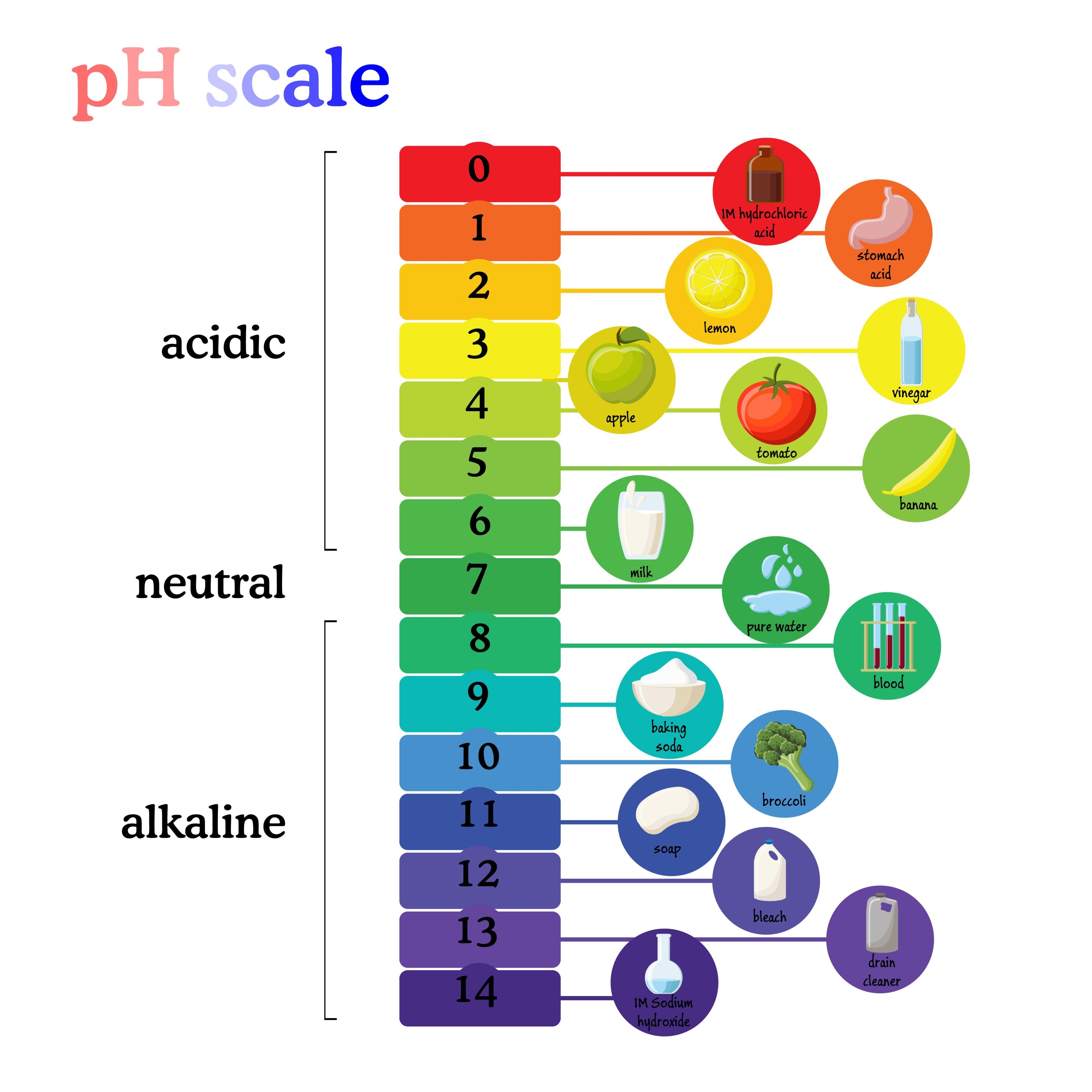
Q5. What is the pH value of pure water?
(a) 0
(b) 7
(c) 14
(d) 1
Answer: (b) 7
Logic:
A pH value of 7 indicates a neutral substance. Values less than 7 are acidic, while values greater than 7 are basic or alkaline.
Q6. Which acid is found in ‘Ant Stings’?
(a) Acetic Acid
(b) Citric Acid
(c) Formic Acid (Methanoic Acid)
(d) Lactic Acid
Answer: (c) Formic Acid
Logic:
When an ant bites, it injects Formic Acid, which causes pain and irritation. Vinegar contains acetic acid, while curd contains lactic acid.
Q7. ‘Bronze’ is an alloy of which two metals?
(a) Copper and Zinc
(b) Copper and Tin
(c) Iron and Carbon
(d) Lead and Tin
Answer: (b) Copper and Tin
Logic:
Bronze is formed by mixing Copper and Tin. Copper and Zinc form Brass. Questions related to alloys are frequently asked in RRB exams.
Q8. Which of the following is used as a moderator in Nuclear Reactors?
(a) Ordinary Water
(b) Heavy Water (D₂O)
(c) Liquid Sodium
(d) Hydrogen
Answer: (b) Heavy Water (D₂O)
Logic:
Heavy water is used to slow down fast-moving neutrons during nuclear fission, helping to control the nuclear reaction efficiently.
Q9. The hardest natural substance on Earth is:
(a) Gold
(b) Iron
(c) Diamond
(d) Platinum
Answer: (c) Diamond
Logic:
Diamond is an allotrope of carbon and is the hardest known natural substance due to its strong three-dimensional crystal structure.
Q10. Which gas is filled in electric bulbs to prevent the filament from burning?
(a) Oxygen
(b) Hydrogen
(c) Argon
(d) Chlorine
Answer: (c) Argon
Logic:
Argon is an inert gas and does not react easily. It prevents the tungsten filament inside the bulb from reacting with oxygen and burning out. Nitrogen is also sometimes used.
5 Tips to Prepare Chemistry for RRB Group D
Chemistry is not about memorizing thick books. It is about understanding the logic behind elements and reactions. If you study smartly by focusing on the right charts and tables, you can finish the syllabus quickly. Here are 5 simple tips to help you master Chemistry for the RRB Group D exam:
1. Master the Periodic Table: You don’t need to learn all 118 elements. Just memorize the first 20 elements (Hydrogen to Calcium) and the Noble Gases (Group 18). Questions on Atomic Number and Valency come directly from here.
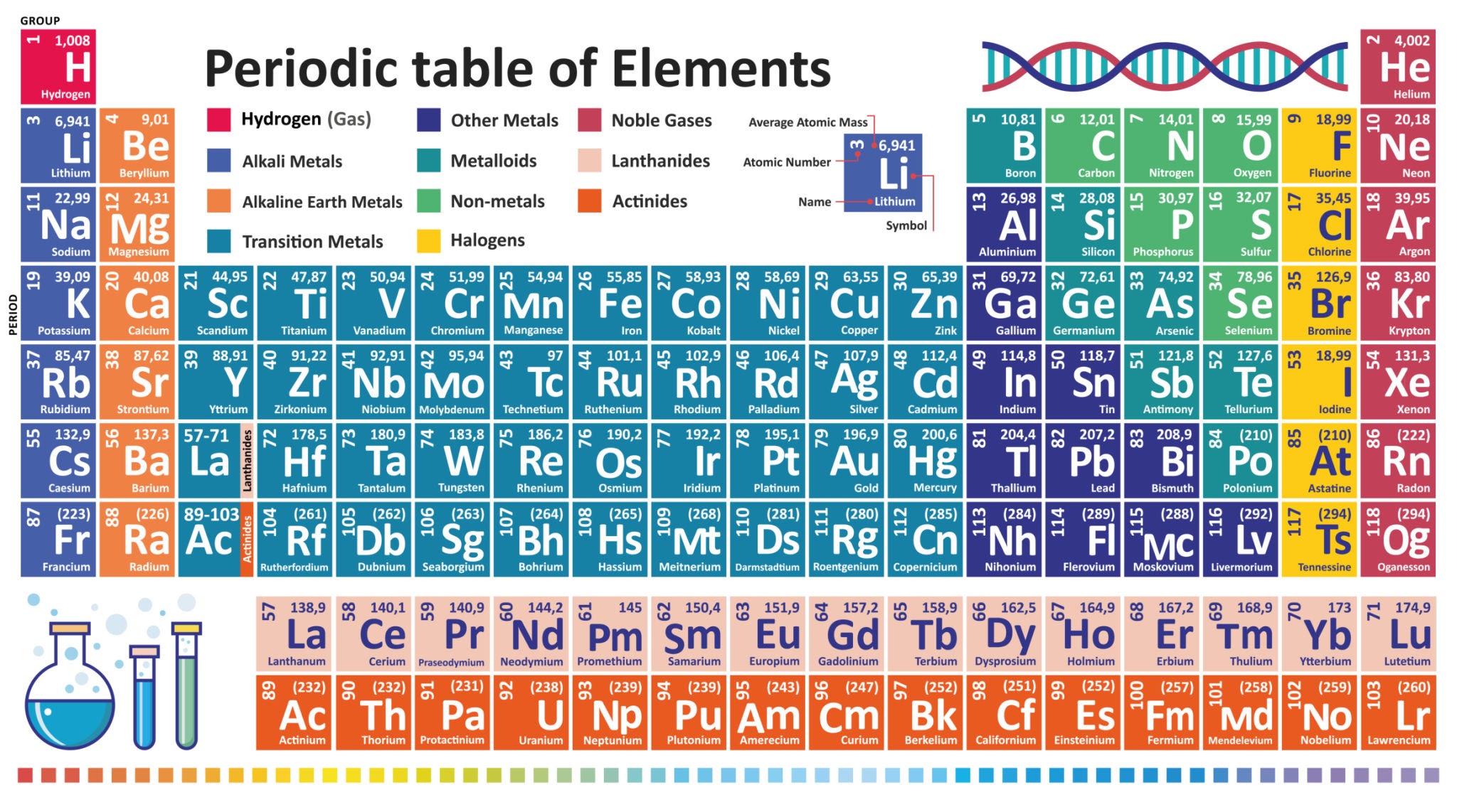
2. Learn Chemical Names: RRB loves asking “Common Names” vs. “Chemical Names.” Make a list:
- Common Salt = Sodium Chloride
- Bleaching Powder = Calcium Oxychloride
- Vinegar = Acetic Acid
3. Understand the pH Scale: Remember the pH values of common things like Lemon (2.5), Blood (7.4), and Water (7). Know that Acid turns Blue Litmus Red.
4. Focus on Alloys & Ores: Memorize which metal comes from which ore (e.g., Bauxite is the ore of Aluminum). Also, learn the composition of Brass, Bronze, and Solder.
5. Attempt Topic Tests: Reading is passive. Testing is active. Attempt our Free Topic Tests to check if you remember the formulas under time pressure.
Join our exclusive Telegram group for expert guidance, personalized tips, and real-time solutions to boost your SSC exam prep. [Click here to join now!]
FAQs: Chemistry Questions for RRB Group D
No. The level is strictly 10th standard (NCERT). If you know the basics of Acids, Bases, and Metals, you can score well.
You can expect around 8 to 10 questions in the General Science section.
Very rarely. Sometimes a simple question on “Mole Concept” or “Atomic Mass” might appear, but 95% of questions are theory-based.
You can download the free, exam-level PDF by clicking the link provided in this blog post.
- Sign Up on Practicemock for Updated Current Affairs, Topic Tests and Mini Mocks
- Sign Up Here to Download Free Study Material
Free Mock Tests for the Upcoming Exams
- IBPS PO Free Mock Test
- RBI Grade B Free Mock Test
- IBPS SO Free Mock Test
- NABARD Grade A Free Mock Test
- SSC CGL Free Mock Test
- IBPS Clerk Free Mock Test
- IBPS RRB PO Free Mock Test
- IBPS RRB Clerk Free Mock Test
- RRB NTPC Free Mock Test
- SSC MTS Free Mock Test
- SSC Stenographer Free Mock Test
- GATE Mechanical Free Mock Test
- GATE Civil Free Mock Test
- RRB ALP Free Mock Test
- SSC CPO Free Mock Test
- AFCAT Free Mock Test
- SEBI Grade A Free Mock Test
- IFSCA Grade A Free Mock Test
- RRB JE Free Mock Test
- Free Banking Live Test
- Free SSC Live Test




